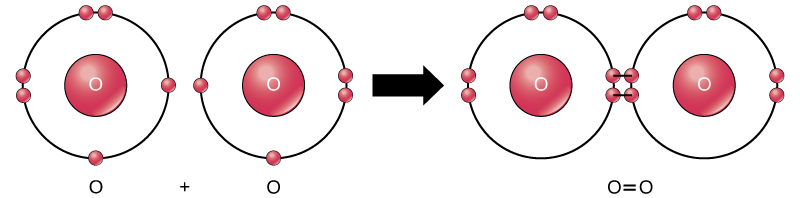
In Chapter 9, we ll look at the transition to the first excited state in formaldehyde. The orbitals suggest that we ll find very different frequencies for the two systems. In Chapter 4, we ll examine the IR spectra for these two molecules. Of the six valence electrons on the oxygen atom, two are involved in the double bond with the carbon, and the other four exist as two lone pairs. 
This difference is due to the two lone pairs on the oxygen. 2,3-Diphenylthiirene 1-oxide and 1,1-dioxide show absorption due to the 1,2- diphenyl-ethylene chromophore. The lowest excited states of thiirane 1- oxide involve excitations from the two lone pairs of the oxygen atom (79G19). The latter shows a positive Cotton effect at low energy followed by a negative effect at high energy. The UV spectra of thiirane 1-oxide and (15,25)-(+)-2-methylthiirane 1-oxide show a broad maximum at about 205 nm (e -23 000). Efftcientiy cooled ozone generators with typical short residence times (seconds) can operate near ambient temperature where thermal decomposition is small. The reaction O3 + 0( P) - 2 O2, is exothermic and can contribute significantly to heat evolution. Processes involving ions such as 0/, 0/, 0, 0, and 0/ are of minor importance. Rate constants for these reactions are given ia References 11 and 93. Ozone can be destroyed thermally, by electron impact, by reaction with oxygen atoms, and by reaction with electronically and vibrationaHy excited oxygen molecules (90). Laser emission at 5 p.m has been obtained from the reaction of methylene and oxygen initiated by flash photolysis of a mixture of SO2, 2 2 6 (1 ). When such reactions are carried out at very low pressure (13 mPa (lO " torr)), energy transfer is diminished, as with molecular beam experiments, so that the distribution of vibrational and rotational energies in the products can be discerned (189). Examples include reactions of oxygen atoms with carbon disulfide (190), acetylene (191), or methylene (190), all of which produce emission from vibrationaHy excited carbon monoxide. Įlectronic excitation from atom-transfer reactions appears to be relatively uncommon, with most such reactions producing chemiluminescence from vibrationaHy excited ground states (188-191). These are either combination reactions or atom- transfer reactions involving transfer of chlorine (184) or oxygen atoms (181,185-187), the latter giving excited oxides. Ī number of chemiluminescent reactions have been studied by producing key reactants through pulsed electric discharge, by microwave dissociation, or by observing the reactions of atoms and free radicals produced in the inner cone of a laminar flame as they diffuse into the flame s cool outer cone (182,183). The collision dynamics of this very reactive state have also been studied in crossed- beam reactions with a RF discharge source which has been. 
The first excited electronic state, 0( D), has a different spin multiplicity than the ground 0( P) state and is electronically metastable. Thennal dissociation is not suitable for the generation of beams of oxygen atoms, and RF and microwave discharges have been employed in this case. An example is the fonnation of oxygen atoms in the upper atmosphere by predissociation of electronically excited O2 molecules. In chemistry, an important class of monomolecular reactions is the predissociation of metastable (excited) species. Classic examples are the spontaneous emission of light or spontaneous radioactive decay.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
